 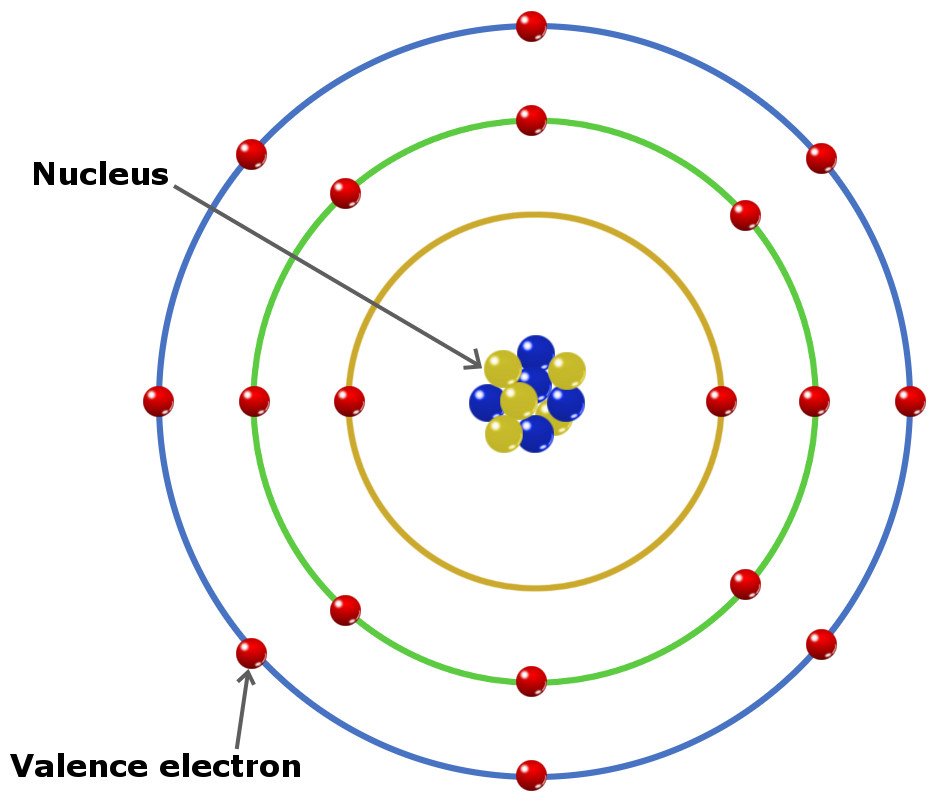 
However, the energy levels of transition elements are (n-1)d, which are approximately close to the ns energy level. This configuration is sometimes expressed in part as Ne for neon (a noble gas), where Ne refers to the core electrons. Another way to show its electron configuration is the short form where its core electrons have the same electronic configuration as this: 3s 2 3p 3. The electronic configuration of phosphorus (P) is 1s 2 2s 2 2p 6 3s 2 3p 3 (long form). In this regard, the quantity of valence electrons a molecule may possess is directly significant and also a function of its electron configuration. The valence electrons of a main-group element are those located in the electronic shell with the largest primary quantum number, n. The highest energy electrons not only determine its valence but also help in determining the chemical behavior of an atom. Valence Electrons and Electronic ConfigurationĪn electronic configuration of an atom refers to the arrangement of electrons in the shells. Nitrogen, on the other hand, may form NH 3, hence it has a valence of 3 and three valence electrons. Historically, the valence of an element was determined by the number of hydrogen atoms it could connect to (which is determined by the number of valence electrons it has available for bonding): for instance, carbon can create CH 4, thus it has a valence of 4 and four valence electrons. Valence refers to an element’s capacity to establish chemical connections with other atoms. (Talk 2022) Figure 1: Diagram of sodium’s electrons. It is located on the outside shell (in this case, the shell resembles a ring). The periodic table can help to understand and determine the valence electrons number an element (particularly a neutral atom of the element) possesses. This also implies that the quantity of valence electrons an element possesses impacts its reactivity, electronegativity, and bonding capacity.įor instance, the valence electron is illustrated in red, which is the simplified picture of sodium’s electrons shown below.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
